Refer Again to the Graph. In Which Region Is the Enzyme Saturated With Substrate
The issue of substrate concentration on enzyme activity
Skip the theory and become straight to: How to make up one's mind Km and Vmax
A elementary chemic reaction with a single substrate shows a linear relationship betwixt the charge per unit of germination of product and the concentration of substrate, as shown below:
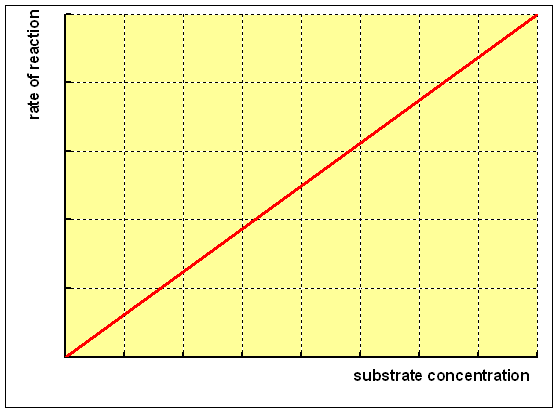
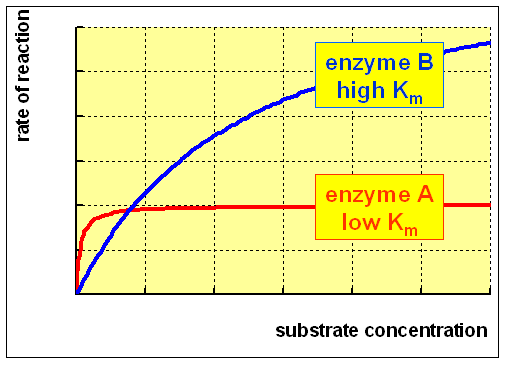
For an enzyme-catalysed reaction, there is commonly a hyperbolic relationship between the rate of reaction and the concentration of substrate, every bit shown below:
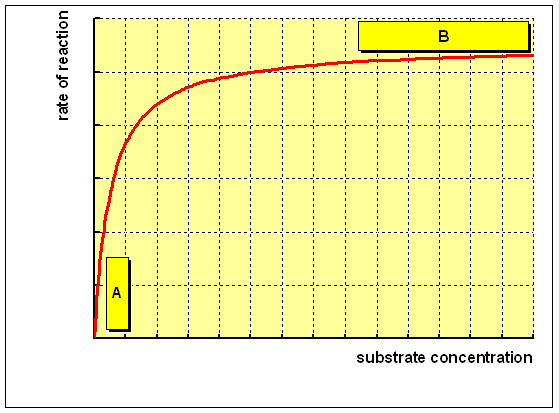
(A) At low concentration of substrate, there is a steep increase in the charge per unit of reaction with increasing substrate concentration. The catalytic site of the enzyme is empty, waiting for substrate to demark, for much of the fourth dimension, and the rate at which product can be formed is limited by the concentration of substrate which is available.
(B) As the concentration of substrate increases, the enzyme becomes saturated with substrate. As soon equally the catalytic site is empty, more than substrate is bachelor to demark and undergo reaction. The rate of formation of product now depends on the activity of the enzyme itself, and adding more substrate will not affect the charge per unit of the reaction to any significant event.

The rate of reaction when the enzyme is saturated with substrate is the maximum rate of reaction, Vmax. For practical purposes, Km is the concentration of substrate which permits the enzyme to reach half Vmax. An enzyme with a high Km has a low affinity for its substrate, and requires a greater concentration of substrate to achieve Vmax."
The relationship betwixt rate of reaction and concentration of substrate depends on the analogousness of the enzyme for its substrate. This is usually expressed equally the Km (Michaelis constant) of the enzyme, an inverse measure of analogousness.
The importance of determining Km and Vmax
The Km of an enzyme, relative to the concentration of its substrate nether normal conditions permits prediction of whether or not the rate of germination of product will be affected by the availability of substrate.
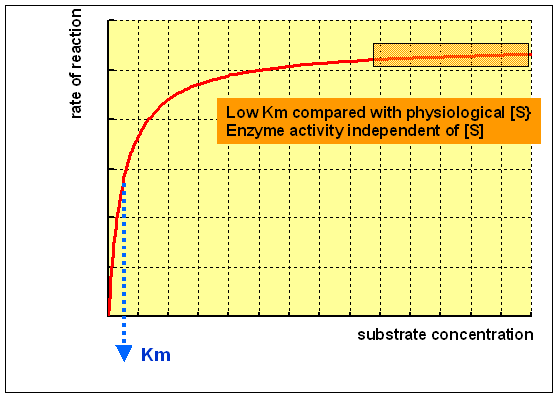
An enzyme with a low Km relative to the physiological concentration of substrate, as shown above, is normally saturated with substrate, and will act at a more or less abiding rate, regardless of variations in the concentration of substrate within the physiological range.
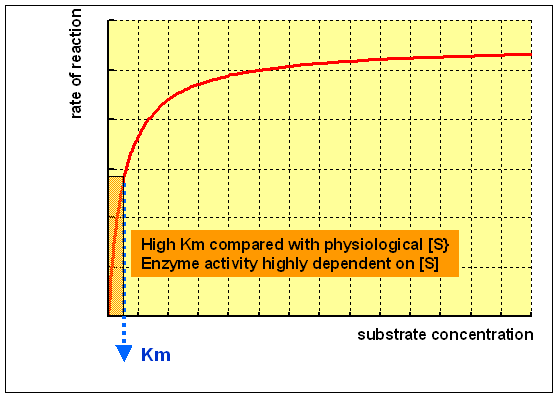
An enzyme with a loftier Km relative to the physiological concentration of substrate, every bit shown in a higher place, is not normally saturated with substrate, and its activity volition vary as the concentration of substrate varies, so that the rate of formation of product will depend on the availability of substrate.
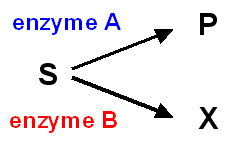 If two enzymes, in different pathways, compete for the same substrate, then knowing the values of Km and Vmax for both enzymes permits prediction of the metabolic fate of the substrate and the relative amount that volition catamenia through each pathway under diverse conditions.
If two enzymes, in different pathways, compete for the same substrate, then knowing the values of Km and Vmax for both enzymes permits prediction of the metabolic fate of the substrate and the relative amount that volition catamenia through each pathway under diverse conditions.
In order to determine the amount of an enzyme present in a sample of tissue, information technology is evidently essential to ensure that the limiting factor is the activity of the enzyme itself, and not the amount of substrate available. This means that the concentration of substrate must be high enough to ensure that the enzyme is acting at Vmax. In exercise, it is usual to use a concentration of substrate about 10 - 20-fold higher than the Km in order to make up one's mind the activity of an enzyme in a sample.
If an enzyme is to be used to determine the concentration of substrate in a sample (e.g. glucose oxidase is used to measure plasma glucose), then the substrate must be the limiting factor, and the concentration of substrate must be below Km, so that the rate of germination of product increases steeply with increasing concentration of substrate, so providing a sensitive assay for the substrate."
How to determine Km and Vmax
Km and Vmax are adamant by incubating the enzyme with varying concentrations of substrate; the results can exist plotted as a graph of rate of reaction (v) against concentration of substrate ([S], and will normally yield a hyperbolic curve, equally shown in the graphs above.
The human relationship is defined by the Michaelis-Menten equation:
v = Vmax / (1 + (Km/[Due south]))
I t is difficult to fit the all-time hyperbola through the experimental points, and difficult to determine Vmax with whatever precision by estimating the limit of the hyperbola at infinite substrate concentration. A number of means of re-arranging the Michaelis-Menten equation have been devised to obtain linear relationships which permit more precise fitting to the experimental points, and estimation of the values of Km and Vmax. There are advantages and disadvantages associated with all three main methods of linearising the data.
The Lineweaver-Burk double reciprocal plot rearranges the Michaelis-Menten equation equally:
1 / v = ane / Vmax + Km / Vmax x one / [S]
plotting 1/5 against 1/[S] requite a straight line:
- y intercept = ane / Vmax
- gradient = Km / Vmax
- x intercept = -1/ Km
-
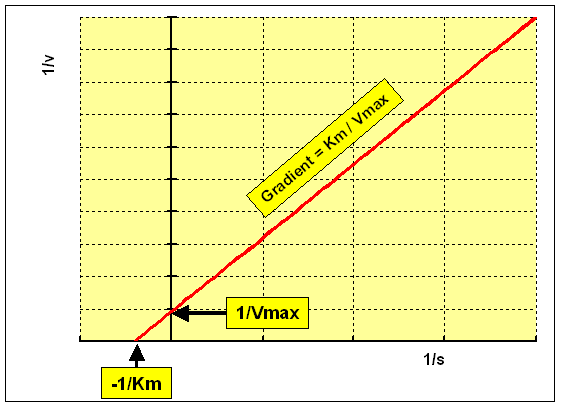
This is the virtually widely used method of linearising the data, and by and large gives the best precision for estimates of Km and Vmax. All the same, it has the disadvantage of placing undue weight on the points obtained at low concentrations of substrate (the highest values of 1/[Due south] and one/v). These are the points at which the precision of determining the rate of reaction is everyman, because the smallest amount of product has been formed.
The Eadie-Hofstee plot rearranges the Michaelis-Menten equation as:
v = Vmax - Km 10 5 / [S]
plotting v confronting v / [South] gives a straight line:
- y intercept = Vmax
- gradient = -Km
- x intercept = Vmax / Km
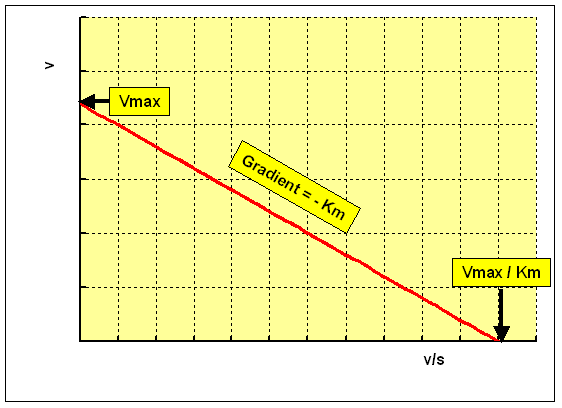
This plot overcomes the problem of uneven spacing of points, and undue weight given to points at depression concentrations of substrate. However, it has the disadvantage that v, which is a dependent variable, is used on both axes, and hence errors in measuring the charge per unit of reaction are multiplied, resulting in lower precision of the estimates of Km and Vmax
The Hanes plot rearranges the Michaelis-Menten equation as:
[S] / five = Km / Vmax + [Due south] / Vmax
plotting [S] / 5 against [Due south] gives a direct line:
- y intercept = Km / Vmax
- gradient = i / Vmax
- 10 intercept = -Km

This plot overcomes the problem of uneven spacing of points, and undue weight given to points at low concentrations of substrate. Yet, information technology has the disadvantage that [S] is used on both axes, and hence pipetting errors, which lead to errors in the true concentration of substrate bachelor, are multiplied, resulting in lower precision of the estimates of Km and Vmax.
Source: https://www.ucl.ac.uk/~ucbcdab/enzass/substrate.htm
0 Response to "Refer Again to the Graph. In Which Region Is the Enzyme Saturated With Substrate"
Post a Comment